Canada's Strategy for Patient-Oriented Research
Improving health outcomes through evidence-informed care
August 2011
Table of Contents
- Preface
- Executive Summary: Canada's Strategy for Patient-Oriented Research
- Introduction
- The Canadian Landscape
- 2.1 The Canadian Bedrock
- 2.2 Two Research-to-Practice Valleys
- 2.3 Key Contributing Factors
- 2.3.1 Decentralized Authority and Priority-Setting
- 2.3.2 Gaps in Human Resource Training and Career Support
- 2.3.3 Financial Constraints
- 2.3.4 Lack of Standardized Data and Technology Platforms
- 2.3.5 Delays in the Approval of Large, Multi-Site Clinical Studies
- 2.3.6 Deficiencies in Guideline Development, Dissemination and Uptake
- 2.3.7 The Limited Role of Patients in Patient-Oriented Research
- The International Landscape
- The Strategy
- 4.1 Vision
- 4.2 Goals
- 4.3 Principles
- 4.4 Bridging the Research-to-Practice Valleys
- 4.4.1 Establishing Collective Leadership
- 4.4.2 Developing the Talent Pool
- 4.4.3 Enhancing the Conduct, Funding and Effectiveness of Multi-Centre Clinical Trials
- 4.4.4 Focusing on Patient-Oriented Research Networks
- 4.4.5 Building and Resourcing SUPPORT Units
- 4.4.6 Improving Guideline Development, Dissemination and Uptake
- Conclusion
- References
- Annex 1: President's Advisory Board Members – Strategy for Patient-Oriented Research
Preface
This document sets out a vision and strategy to improve health outcomes and enhance patient care through the levers of research. The underlying premise of the document is that greater uptake of research-based evidence will improve the health of Canadians while improving the cost-effectiveness of the health care system.
This document was developed under the leadership of the Canadian Institutes of Health Research (CIHR), Canada's premier health research agency. The focus on patient-oriented research is at the core of CIHR's 2009/10 Health Research Roadmap which outlines CIHR's strategic directions over the next five years.
CIHR was guided in this work by a 12-member President's Advisory Board drawn from across Canada and abroad, representing a breadth of experience, expertise and perspectives. The Advisory Board met between January 2009 and August 2010. Its work was ably supported by Dr. Paul Hébert with assistance from Dr. Joe Pater, both to whom a huge debt of gratitude is owed. The Advisory Board also benefited enormously from the advice and assistance of the CIHR's 13 Scientific Directors and staff at many levels of the organization.
Between February and June 2010, a series of consultations were held on an earlier version of this document. In-person meetings were held at universities, with academic health care organizations, life sciences industries, health charities, with health professional associations, and other relevant stakeholders. Meetings were also held with federal government officials from Health Canada, the Department of Finance, the Privy Council Office, the Public Health Agency of Canada and Industry Canada, as well as with officials from provincial and territorial governments and provincial health research funding agencies. In addition, a web-based survey, hosted and analyzed by Harris/Decima, was undertaken. Of the 288 respondents, the overwhelming majority were health researchers with 10% coming from the general public.
The results of these consultations were discussed at the final meeting of the Advisory Board on August 17th, 2010. Overall, there was strong support for the need for a strategy for Canada and for its proposed elements. Numerous suggestions for improvement were provided and gaps identified. Of particular note was the need to better reflect the patient perspective in the Strategy, to recognize and respond to regional differences and needs, and to more clearly address the breadth of health care disciplines and health care settings within the Strategy.
This final document of the Advisory Board responds to this extensive input as well as additional advice from the Advisory Board members and CIHR Scientific Directors. This will now become the foundation document for a National Steering Committee charged with overseeing the development and implementation of an action plan. It is hoped that this document will facilitate the engagement of stakeholder groups and partners and begin to catalyze the changes that are needed to transform the health care landscape in this country through research and innovation for the benefit of all Canadians.
Executive Summary: Canada's Strategy for Patient-Oriented Research
Patient-oriented research, the cornerstone of evidence-informed health care, refers to a continuum of research, from initial studies in humans to comparative effectiveness and outcomes research, and the integration of this research into the health care system and clinical practice.
The goal of patient-oriented research is to better ensure the translation of innovative diagnostic and therapeutic approaches to the point of care, as well as to help the provinces and territories meet the challenge of delivering high quality, cost-effective health care. It involves ensuring that the right patient receives the right clinical intervention at the right time, ultimately leading to better health outcomes.
The vision of the Strategy for Patient-Oriented Research is to demonstrably improve health outcomes and enhance patients' health care experience through integration of evidence at all levels in the health care system.
The goals to achieve this vision are:
- To create a collaborative, pan-Canadian process for identifying, establishing and addressing patient-oriented research priorities;
- To establish an integrated, leading-edge pan-Canadian clinical research infrastructure along the full continuum of patient-oriented research;
- To grow Canada's capacity to attract, train and mentor health care professionals and health researchers, as well as to create sustainable career paths in patient-oriented research;
- To strengthen organizational, regulatory and financial support for clinical studies in Canada and enhance patient and clinician engagement in these studies; and
- To improve processes for the early identification of best practices, expedite their development and harmonization into guidelines for patient care and support their adoption by clinicians, caregivers and patients.
The Problem
Canada already has a strong foundation on which to build excellence in the domain of patient-oriented research; including its basic biomedical research community; its clinical workforce distributed across the country and across disciplines; a highly qualified, internationally respected clinical and health services research workforce; and, world-leading expertise in systematic reviews. Large provincial and national administrative and clinical databases have enabled researchers to assess existing care patterns, make important associations between interventions and outcomes, and observe practice variations. Canada also has extensive experience in multidisciplinary and cross-disciplinary clinical research networks.
Despite these undisputable strengths, Canada faces a dual challenge in the research-to-practice continuum, often referred to as the two "Death Valleys" in the health care landscape. Valley 1 refers to the decreased capacity to translate the results of discoveries generated by basic biomedical research in the laboratory to the bedside or careside as well as to successfully commercialize health discoveries. This negatively impacts Canada's clinical research and knowledge base and its international competitiveness. Valley 2 refers to the limited capacity to synthesize, disseminate and integrate research results more broadly into clinical practice and health care decision-making. These two valleys must be bridged if Canada is to bring evidence to bear to enhance health outcomes and ensure a sustainable health care system.
A number of factors contribute to the gaps in the research-to-practice continuum, including:
- Lack of clinical investigators, methodologists such as biostatisticians, clinical epidemiologists and health economists, and other needed experts;
- Complicated coordination of large, multi-centre clinical trials because of requirements for multiple ethics reviews by each individual institution and the lack of standardized contracts;
- Underfunding of patient-oriented research when compared to many other countries;
- A decentralized federation structure, making research coordination difficult;
- Deficiencies in the development and dissemination of guidelines and their adoption into practice, despite their proven value; and
- The limited role of patients in patient-oriented research.
The Proposed Solution
Collective guidance and leadership for the Strategy from all levels of government is essential to meeting the Strategy's goals and effecting change. The first step proposed for moving forward is the establishment of a national Steering Committee, comprised of representatives from all major stakeholders from public (federal and provincial) and private sectors. The Committee will be tasked with implementing the Strategy and encouraging collective engagement and ownership. The Steering Committee will be guided in its work by the Strategy's seven principles: collaboration; responsiveness; excellence; inclusivity; innovation; accountability and transparency. A key factor is the important role of the patient to ensure that research priorities are relevant and that the transfer of knowledge is effective.
Specific initiatives in five broad areas are proposed to meet the goals of the Strategy and to begin to address gaps in the research-to-practice continuum.
1. Developing the Talent Pool
To build capacity in patient-oriented research, relevant expertise must be developed and fostered. To do so, Canada must re-design career training and salary awards for all types of clinical investigators. Awards will be designed to consider different practice plans and remuneration strategies, and will take into account the institutional commitment to the applicant and their program context. The institutional commitment will include provisions for salary support, recruitment of team members, program development and infrastructure.
Canada also needs more methodologists to support patient-oriented research and more health care professionals, especially those with advanced degrees, trained in the core methods necessary to carry out research activities. This will require universities nation-wide to better support master's and doctoral programs in such areas as epidemiology and biostatistics, health economics and other health-related social sciences. It also implies developing standardized training for research coordinators, project managers and other support personnel.
2. Enhancing the Conduct, Funding and Effectiveness of Multi-Centre Clinical Trials
Designing, implementing and funding multi-centre clinical trials is very difficult in the current Canadian context and Canada is losing its competitive hold. To improve our competitiveness in conducting clinical trials, it is essential to overcome a number of identified barriers such as inadequate funding for trials, the lack of a common contract template to facilitate research between researchers and industry, and the lack of harmonized ethics processes that permit timely and expedited ethics review of multi-centre trials. Of particular importance is the requirement to significantly increase the funding available to support well-designed research studies that address issues important to patients and their care.
3. Focusing on Patient-Oriented Research Networks
The co-ordination of patient-oriented research must be improved. To build on Canada's experience in multidisciplinary or transdisciplinary solutions-focused networks and respond to current challenges with respect to sustainability, linkage and methodological support, the Strategy proposes to support thematic, patient-oriented research networks.
These clinical research networks will identify research priorities and clinical protocols, assist in the scientific oversight of research programs, support scientists through various teaching and mentorship activities, and foster interdisciplinary research. Some of these networks would provide national platforms to undertake clinical trials in specific content areas. Each platform would include development and maintenance of electronic databases as well as relevant data repositories (e.g. biobanking), centralized randomization generation, support for patient recruitment, data collection and data analysis capacity, and facilities to support research ethics board submissions.
4. Building and Resourcing Support Units
To begin to address the need for Canadian capacity related to patient-oriented research and to provide methodological support to those already engaged in or wishing to be engaged in patient-oriented research projects, specialized research service centres referred to as Support for People and Patient-Oriented Research and Trials (SUPPORT) units will be created. SUPPORT units are multidisciplinary centres of methodologists including epidemiologists, biostatisticians, social scientists, health economists and clinical trialists as well as research support staff to assist investigators design relevant research studies, conduct biostatistical analyses, manage data and biobanks, learn project management skills and meet all relevant regulatory standards.
5. Improving Guidelines Development, Dissemination and Uptake
The final element of the Strategy proposes a concerted, well-resourced, collective approach to improve quality of care and health outcomes through the implementation of clinical practice guidelines and best practices. The Strategy aims to facilitate guideline development, dissemination and uptake by increasing capacity for the synthesis of knowledge, providing relevant training to guideline developers, improving understanding of the barriers to implementation, and encouraging guideline implementation and evaluation.
Patient involvement in all aspects of the Strategy is important to achieve desired outcomes.
Conclusion
By 2025, health outcomes in Canada will be demonstrably improved and the health care experience for patients enhanced through the integration of evidence at all levels of the health care system. This is the vision driving the development of Canada's Strategy for Patient-Oriented Research. The Strategy provides value for all stakeholders in the field, including the patient receiving the right care, in the right place at the right time, provincial governments and health care administrators having a cost-effective, efficient, affordable health care system that meets the needs of patients, and the Government of Canada meeting a number of key objectives of the Science and Technology Strategy, including the opportunity to work with partners to enhance the commercialization of research.
1.0 Introduction
1.1 What is Patient-Oriented Research?
Research focused on patients is the cornerstone of evidence-informed health care, a relatively recent concept of clinical care that was largely pioneered in Canada.1 A recent survey undertaken by the British Medical Journal of its readers placed evidence-based medicine as one of the top 10 medical advances of all time.2
Health care professionals and policy makers need evidence to deliver high quality, appropriate and cost-effective care. This requires the timely and efficient translation of research innovations from the research setting to patient care settings. It also calls for the evaluation and synthesis of existing knowledge and rapid transfer of what is already known to the clinical setting. For instance, the use of a simple 19-item checklist developed by the World Health Organization has significantly decreased deaths and complications in routine elective surgeries in Canada and around the world.3
Patient-oriented research refers to a continuum of research, from the initial studies in humans to comparative effectiveness and outcomes research, and the integration of this research into the health care system and clinical practice. It includes studies of therapies, diagnostic tools, pharmaceuticals, and prevention strategies as well as comparative evaluations of those interventions against each other and against existing practices. It concludes with the synthesis, dissemination and integration of evidence into the health care system and clinical practice. Patient-oriented research thus encompasses both clinical research and health services research, the synthesis, dissemination and integration of this new knowledge into the health care system and into clinical practice.
Patient-oriented research focuses on the care of patients in the health care system as opposed to research focused on whole populations, although there is little question that health outcomes are a function, in part, of social determinants and that there are strong synergies between patient-oriented and population health research. This fact speaks to the critical need to continue to advance population health research in Canada and to create strong bridges between researchers in these two fields.
While patient-oriented research begins where basic biomedical research and pre-clinical studies end, both are equally essential and necessarily complementary. In fact, the flow between basic biomedical and patient-oriented research is bi-directional, as observations from patient-oriented research can generate new questions for basic biomedical research.
While a major focus of patient-oriented research is on new discoveries, an equally important focus is comparing new discoveries with existing therapies (Figure 1, right box) to determine which interventions are most beneficial as part of usual care. This "comparative effectiveness research" complements studies designed to demonstrate the efficacy and safety of new drugs or devices prior to their initial use in clinical practice (Figure 1, left box).
Patient-oriented research can be conducted by clinical researchers from all health care professions (e.g. nursing, medicine, dentistry, chiropractic, pharmacy and physiotherapy) by clinical epidemiologists and as well as by non-clinicians (e.g. biostatisticians, health economists, anthropologists, sociologists and behavioural scientists). Patient-oriented research can be undertaken in all health care settings: primary care, tertiary care, long-term care, home care or community-based care, wherever patients seek and/or receive care.
The relatively new nomenclature of "patient-oriented" research connotes a higher level of engagement of the patient in the clinical research and knowledge translation process. Indeed, there is growing support for the view that health research needs to assess interventions and outcomes considered important by patients and clinicians and that by doing so costly mismatches of research-to-needs can be avoided. The valuable input of patient groups is starkly illustrated by their contribution to the rapid development of antiviral therapies for HIV.4
Figure 1: Processes for Developing versus Comparing Clinical Interventions
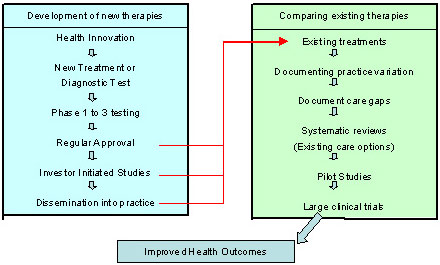
Figure 1 outlines the chain of steps for proving that a drug or device saves lives or decreases morbidity. The step-wise process often requires 15 to 20 years for new interventions and 5 to 7 years if comparing existing therapies.
1.2 Why is a Strategy for Patient-Oriented Research important now?
While clinical research has provided the underpinnings for the development and application of health interventions, comparative evaluations of these interventions that provide guidance as to when and to which patients to apply them have lagged. Additionally, a number of factors in the Canadian environment have added impetus to the need to move forward on the development and implementation of a comprehensive, national strategy for patient-oriented research.
First, while investments in health research have led to the development of a vast array of preventive, diagnostic and treatment interventions for health, there is an increasing impatience among clinicians, policy makers and patients with the pace at which scientific discovery is resulting in new products or interventions. Many feel that progress towards cures and better treatments are not as rapid as they would like, especially for diseases such as cancer, mental health, Alzheimer's and other neurological disorders. In addition, the application of interventions often varies dramatically among and between different health care professionals, institutions and jurisdictions.5
Second, significant gaps in high quality evidence on comparative effectiveness mean that it is difficult to establish guidelines for appropriate care. This lack of evidence is now seen as one of the major drivers for variations in care.6-11 Indeed, several studies have demonstrated that less than 60% of bedside decisions on general medical services have the support of the required level of evidence12-14 and that up to 25% of patients get care that is not needed or could be potentially harmful).15 We also know that patients treated in institutions that are actively involved in research have better health outcomes.16,17 Patients and health care professionals have a right to expect that important health decisions are made on the basis of solid evidence.
Third, in tighter economic times, funders of basic biomedical research, including federal and provincial governments and health charities, are anxious to see and to explain to taxpayers and donors the public benefit of the billions of dollars invested in scientific research. Provincial governments faced with shrinking coffers and rising health care costs are anxious not only to see new discoveries brought to market, but also to benefit from comparisons of new discoveries with existing therapies. These findings would provide information on outcomes and cost-effectiveness so that resources may be allocated to the most effective interventions and diagnostic procedures, potentially resulting in important economic dividends. Indeed, patient-oriented research is not only about doing things better but also about being able to stop doing things that are known to be ineffective, unnecessary and often costly.
Fourth, while sustained government investments in health research over the past decade have yielded many advances and have enabled Canadian researchers to obtain global recognition for their work, many health challenges persist and have proven to be immune to existing approaches. At the same time, new health challenges continue to emerge. The aging of the population is giving rise to increasing co-morbidities in patients resulting in growing concern that new approaches are needed, and needed quickly, to address issues related to chronic disease management. There are significant sub-populations in large metropolitan areas as well as isolated populations in northern, rural and remote regions whose health care needs pose additional challenges and are not optimally met. The Canadian health consumer is also becoming more knowledgeable about his/her health care and has access to enormous amounts of information, both evidence-based and otherwise, creating growing pressure and expectations for results. The increasing focus on self-care and the promise of personalized medicine adds to the sense of urgency to better apply our health care resources.
Canada has not by any means been standing still with respect to patient-oriented research. But the compelling financial imperative in all provinces and territories, both entrenched and emerging health challenges and increasing expectations from providers and patients alike speak to the need to develop a fulsome, collective strategy to move forward. Canada needs to start now as demonstrating research impact is not a simple task, often taking years. This time lag is further amplified by Canada's geography, low population density, especially in the North, its multi-ethnic character and its complex federal/provincial/territorial structure. Thus, there is urgency in beginning now with a thoughtful, collective strategy.
Fortunately, Canada already has the core underlying strengths to build on and broad acceptance that we can do better. There are also many models, both within Canada and internationally, that we can learn from and adapt to different contexts. In short, the need is great, the potential benefits are huge and Canada is ready.
2.0 The Canadian Landscape
2.1 The Canadian Bedrock
Canada's high quality public health care system provides access to all Canadians to health care. Since "usual care" is the comparator in clinical studies, high quality and uniform care is a desirable foundation for patient-oriented research.
Canada's public university system in all parts of the country has produced a strong basic biomedical research community which provides the essential raw material for translation and which has a long tradition of collaborating effectively with clinical researchers.
Canada's dedicated clinical workforce is distributed across the country in academic health centres, regional health authorities, community hospitals and health centres, public health units, primary care practices, nursing homes, schools and government agencies.
Canada's highly qualified clinical and health services research workforce is distributed across the country in this nation's universities, 100 teaching hospitals and other centres and across many disciplines. While it tends to be highly concentrated in tertiary care settings it is also found in primary care and community-based settings. It includes clinicians and other health practitioners engaged in research as well as biostatisticians, health economists, epidemiologists, social scientists (e.g. psychologists, sociologists, health service researchers). It also includes research nurses, research assistants and research support staff.
Canadian clinicians and applied health researchers have been able to conduct and participate in ground-breaking studies and have achieved breakthrough publications in many fields, most notably in cardiovascular care,18,19 critical care,20 stroke,21 HIV,22 care of pre-term infants,23 pain management,24 and oncology25. Clinical research networks organized along thematic lines (e.g. the National Cancer Institute of Canada Clinical Trials Group,26 the HIV Clinical Trials Group27 and the Canadian Critical Care Trials Group28 have led to publications in high-profile international journals. In fact, Canada's clinical researchers have been "punching above their weight" for some time internationally, as evidenced by their citations in academic journals (Figure 2).
Figure 2. Canada's Scientific Impact in the Health Field in the International Context
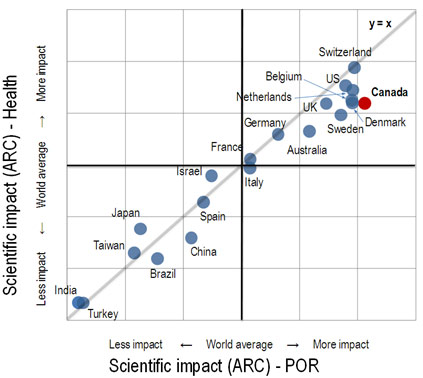
Source: Science-Metrix, Patient-Oriented Research in Canada: A Bibliometric Analysis. 2009.
Figure 2 illustrates Canada's scientific impact in the health field in the international context. In 2009, Canada ranked highly, together with the United Kingdom, Belgium, the Netherlands, Denmark and the United States in scientific impact for the health sector as a whole (ordinate), but ranked first in patient-oriented research (POR abscissa).
Canada's expertise in systematic reviews has garnered recognition internationally. Canadian experts not only conduct a disproportionally large number of major systematic reviews and meta-analyses, but they have also been instrumental in developing the science underpinning this important element of research. Systematic reviews are an essential building block for evidence-informed practice and the development of clinical practice guidelines and a necessary step in the planning process of large comparative trials. They are now the most frequently cited form of clinical research.29
Canada was an early participant in and builder of the Cochrane Collaboration, an international, non-profit network helping health care providers, policy makers, patients and caregivers make better informed decisions about health care by preparing, updating and promoting the accessibility of systematic reviews. Canada's contributions include the CIHR-funded Canadian Cochrane Centre, including the Cochrane Effective Practice and Organization of Care Group that conducts systematic reviews of the effectiveness of interventions to influence professional practice.
The Canadian Agency for Drugs and Technologies in Health (CADTH), a comprehensive, pan-Canadian health technology assessment agency, supports evidence-informed decision-making by providing health technology assessments and other tools to promote the optimal use of drugs, devices and other health technologies, based on its own systematic reviews and meta-analyses.
The Canadian Task Force on Preventive Health Care, re-established in 2010 by the Public Health Agency of Canada (PHAC), develops and disseminates clinical practice guidelines for primary and preventive care based on systematic reviews of the scientific evidence. CIHR and PHAC recently funded an Evidence Synthesis Centre out of McMaster University to support the knowledge synthesis needs of the Task Force.
Canada's large provincial and national administrative and clinical databases, such as the Ontario Health Insurance Plan database and the Canadian Institute for Health Information (CIHI) database, enable researchers to assess existing care patterns, make important associations between interventions and outcomes, and observe practice variations. Centres such as the Institute of Clinical Evaluative Sciences (ICES) in Ontario, the Manitoba Centre for Health Policy and the British Columbia Linked Health Database have the ability to anonymously link population health information on an individual patient basis using unique identifiers that ensure the confidentiality of the information. They are thus able to obtain a more comprehensive view of specific health care issues than can be achieved with unlinked data. This makes it possible to identify gaps in health care, particularly in the use of treatment options, and highlight drug safety issues and variations in practice patterns.
New investment in the Drug Safety and Effectiveness Network, a collaboration between CIHR, Health Canada and stakeholders across the country, introduces post-market monitoring and evaluation of drugs and is a welcome step in extending the evidence base across the continuum of care.
Recent federal investments through Canada Health Infoway are assisting provincial governments to accelerate the development of electronic health records. Electronic health records are essential to delivering patient-centred care. They have the potential to reduce the number of duplicate tests, improve diagnoses, enable more collaborative approaches between physicians, nurses and other health care providers, provide more efficient and comprehensive care and ultimately improve health outcomes. Unfortunately, progress has been slower than hoped with respect to the uptake by provinces of electronic health records. Currently, about half of Canadians have an electronic medical record stored in electronic form but less than 20% have an electronic health record that links all of their disparate health information and that is accessible to all health professionals.30
Canada's extensive experience in developing and working in clinical networks has laid a solid foundation on which to build. The Government of Canada's twenty-year old Networks of Centres of Excellence (NCE) Program is hailed internationally as a precedent-setting network model in which barriers between disciplines, institutions and sectors are broken down. The Program builds in operational funding for the networks as well as a requirement for knowledge translation including commercialization of research. It also includes an expectation on networks to ensure their sustainability after the federal funds have ceased, although remarkably few of the NCE's have been successful at meeting this sustainability criteria. A number of the health-related research networks such as the Canadian Stroke Network, the Arthritis Network, and the Stem Cell Network received the full 14 years of government funding and are broadly regarded as having had a significant impact in their fields.
An environmental scan recently undertaken for CIHR on clinical research networks in Canada31 noted an array of such networks across Canada, predominately national in scope and largely disease-based in approach. The most common types were "focused networks" pursuing a solution to a specific health problem; "networks for clinical trials" and "quality assurance networks" focused on changing provider behaviour. By far the most common source of funding identified for these networks was research grant funding. The scan noted that these networks commonly had a strategic plan but were less likely to have protocols for data-sharing, intellectual property and publication. Scientific priorities dominated the approach to priority-setting for the networks as well as funder's needs, with it being much less common to include patients in the priority-setting process. The main issue cited by the clinical research networks in the scan was sustainability.
Numerous other networks have grown informally across the country. These informal networks are often "bootstrapped" from individual research grants and by the strong will researchers have to work together. They thus can link to each other only when funds permit and tend to disband when funding runs out. For example, some 120 informal networks alone were noted in the field of child and youth health. The establishment of the Maternal, Infant, Child & Youth Network (MICYRN) in 2006, was a collaborative effort to bring networks together in this field. Its primary objectives are to build innovative platforms to address gaps in the clinical research environment and enhance partnerships, training, knowledge translation and research methods. It also set out to establish integrated access to innovative tools in data management, genomics and biobanking.
The Government of Canada's Science and Technology Strategy, announced in 2007, provides an important framework for a patient-oriented research strategy with its emphasis on building research excellence, translating knowledge into practical applications, reducing barriers and deepening the pool of highly skilled individuals.
Recent efforts to enhance coherence in Canada's health research enterprise by health research funders including CIHR, the provincial health research organizations, academic health care organizations, and the health charities have improved data sharing and streamlined processes. These organizations are committed to continuing to harmonize processes and address inefficiencies that have plagued clinical research in the past.
2.2 Two Research-to-Practice Valleys
While Canada has a strong bedrock on which to build, a number of factors have combined to impede the flow of information from basic biomedical research through clinical research to changes in patient care and improved health outcomes.
In effect, Canada faces a dual challenge in the research-to-practice continuum, often referred to as the two "Death Valleys" (Figure 3). Valley 1 refers to the decreased capacity to translate the results of discoveries generated by basic biomedical research in the laboratory to the bedside or careside as well as to successfully commercialize health discoveries. This negatively impacts Canada's clinical research and knowledge base and its international competitiveness. Valley 2 refers to the limited capacity to synthesize, disseminate and integrate research results more broadly into health care decision-making and clinical practice. These two valleys must be bridged if Canada is to bring evidence to bear to enhance health outcomes and ensure a sustainable health care system.32
Figure 3: The Two Valleys of the Research-to-Practice Continuum

Figure adapted from Steven Reis, University of Pittsburgh and Harold Pincus, Columbia University.
2.3 Key Contributing Factors
2.3.1 Decentralized Authority and Priority-Setting
Canada's decentralized federation structure presents challenges for the leadership and coordination of patient-oriented research.
Provincial and federal jurisdictions have distinct, as well as overlapping, responsibilities for the funding and regulation of health care and health research.
While health is primarily a matter of provincial responsibility, the federal government has a number of important roles. It has regulatory authority over a wide array of pharmaceutical, therapeutic, food and consumer products and many aspects of assisted human reproduction, setting safety, efficacy and quality standards. It is a provider of health care services to specific 'federal populations', most notably, First Nations and Inuit, but also veterans, prisoners in federal institutions and members of the Department of National Defence and the RCMP. It is a partner with provinces and territories in the development and sustainability of Canada's universal public health care system through the Canada Health Transfer and the Canada Health Act and through financial support to a number of national health organizations including CIHI, Canada Health Infoway, CADTH, the Health Council, the Mental Health Commission, the Patient Safety Institute and the Partnership Against Cancer. It has a recognized role in public health and health promotion mainly through the Public Health Agency of Canada, but also through programs of Health Canada ranging from emergency health preparedness, to chronic and infectious disease management strategies, public health surveillance, and regulatory and tax measures to influence health behaviours.
The provinces are responsible for planning and financing most health care services and support the major institutions where patient-oriented research is conducted. They produce the clinicians and the researchers who are at the core of patient-oriented research as well as the health care providers and administrators who are the recipients of the new knowledge and translate it into advances in health care and who are at the forefront of interactions with patients and their families.
Both jurisdictions have invested significantly in health research. Federal investments have primarily been through CIHR but also range across a number of programs and agencies such as Genome Canada, the Canada Foundation for Innovation (CFI), the Canadian Health Services Research Foundation (CHSRF), the Canada Graduate Scholarships, the Canada Research Chairs Program, the Indirect Costs of Research Program and, most recently, the Vanier Canada Graduate Scholarships and the Banting Postdoctoral Fellowship. The provincial governments fund health research directly through their provincial health research funding agencies and, indirectly, through their investments in the universities and academic health care centres where health research is undertaken. They have also been frequent co-funders of specific initiatives under CFI and CHSRF.
This jurisdictional reality, while fruitful at one level, can make for suboptimal coordination of patient-oriented research leading to inefficiencies and under-performance. Most significantly, this structure has meant the absence of pan-Canadian standards as well as barriers to the sharing of patient and utilization data across provinces. Pan-Canadian standards are critical because research increasingly crosses provincial boundaries. The scattered approach to the development and implementation of clinical practice guidelines and the lengthy process for ethic reviews for large, multi-site clinical trials are examples of the impact of these jurisdictional differences.
Additionally, federal and provincial patient-oriented research investments at present do not reflect an overall strategy or established, agreed-upon priorities based on the identified needs and preferences of importance to Canadians. Nor are they based on burden of illness. Overall, public funding of health research is rarely correlated with disease burden, if at all.29 A number of provincial jurisdictions are establishing provincial health research priorities, with an increasing focus on primary care and chronic disease management. But Canada continues to lack a mechanism to bring all relevant players to the table to determine pan-Canadian health research priorities and to work collaboratively to reduce duplication and maximize clinical expertise across the country.
2.3.2 Gaps in Human Resource Training and Career Support
As previously noted, Canada has an impressive number of highly trained and internationally recognized individuals in its clinical research workforce. However, there are significant and growing gaps with respect to clinical investigators and troubling shortages of biostatisticians, health economists, clinical epidemiologists and social scientists focused on clinical research. Leaders in the research community have also highlighted the need for non-clinical research staff and support staff such as trained project coordinators and research assistants.
CIHR's 2002 report "The Clinician Scientist: Yesterday, Today and Tomorrow" notes that clinical investigators of all types find that their research commitment is increasingly difficult to pursue, being frequently overwhelmed by clinical and teaching duties, often as a result or growing shortages of health care professionals in their particular fields. Given their relative lack of time to engage in research, clinical investigators often have difficulty competing in the current environment for career awards.
Some barriers that prevent clinical investigators from initiating or continuing a career in research include:
- Few mentors or successful role models;
- Limited training opportunities;
- Heavy clinical workload;
- Lack of research time protection from division and department heads;
- Poor first experience in research;
- Lack of a team environment;
- Lack of methodological support; and
- Inappropriate compensation.33-38
Despite CIHR and provincial efforts to address this reality, many reports39-44 concur that Canada has failed to adequately deal with this shortage of clinical investigators. On the medical side, the latest national survey of the Canadian Medical Association shows that only 4% to 7% of physicians report participating in research more than 20 hours per week. This proportion has remained constant over the last 10 years despite important increases in health research spending from 2000 to 2007.45
There is no evidence of an increase in the number of trainees and young health care professionals entering research careers supported by either CIHR or provincial organizations. Uptake for CIHR training and salary awards by physician trainees has stayed relatively constant whereas CIHR training and salary award funding for biomedical scientists has increased by more than 50% over the last five years, its training and salary award funding of physicians has shown a downward trend (Figure 4). Similarly, provincial funding of training and salary awards to physicians has not increased over the past five years (Figure 5). Even fewer awards are made to nurses by either CIHR or the provinces, and this category shows a flat five-year trend. Overall, while CIHR and the provinces have invested heavily in the careers of biomedical researchers, health care professionals/clinicians have not experienced the same level of support.
The creation of the Canadian Health Services Research Foundation (CHSRF) by the federal government over a decade ago drove the growth of a cadre of health service researchers in Canada that had been seriously lacking and badly needed. The unique matching funding requirement for the research grants brought the health system research user to the table with the researcher so as to enhance its uptake and translation into practice. This was, in many ways, an exciting "first" in patient-oriented research in Canada. Another innovative initiative of CHSRF's isthe Executive Training for Research Application (EXTRA) program which seeks to develop capacity and leadership to optimize the uptake and use of research evidence in clinical practice and in managing Canadian health care organizations. Unfortunately, the winding down of the endowment of CHSRF has eroded this advancement despite the transfer of the operating grant portion of the Foundation's mandate to CIHR.
Serious concerns also exist with respect to the level of support for biostatisticians and other methodologists. Biostatisticians are essential to patient-oriented research. However, as they tend to be salaried positions within universities, as opposed to being grant-funded, they have been very vulnerable to budget cuts. There are also shortages of health economists and clinical epidemiologists in Canada. With the increasing focus on evidence-informed care, the need for these experts will continue to grow.
Figure 4. Total Training and Salary Awards from CIHR
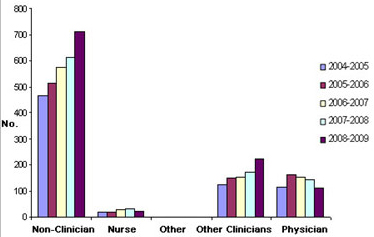
Figure 4 illustrates the total number of annual training and salary awards CIHR awarded to physicians, nurses and other clinical health care professionals from fiscal years 2004-2005 to 2008-2009 with those awarded non-clinician researchers over the same period. Data suggest stagnation in nursing awards, a slight increase in awards to other clinicians, a more substantial increase in awards to non-clinicians and a decline for physicians.
Figure 5. Total Training and Salary Awards from the Provinces
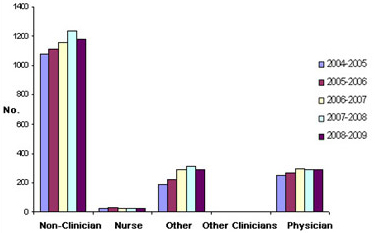
Figure 5 illustrates the total number of annual training and salary awards the reporting provinces (Alberta, British Columbia, Manitoba, Nova Scotia, Ontario and Quebec) awarded to physicians, nurses and other clinical health care professionals with those awarded to non-clinician researchers from fiscal years 2004-2005 to 2008-2009. Data suggest no significant changes in the clinical investigators and a slight recent decrease in those awarded to non-clinicians.
Sources: (1) Provincial data from National Association of Provincial Health Research Organizations (NAPHRO); (2) Canada data from CIHR database (EIS).
This lack of support for health care professionals to develop and maintain research careers is a serious threat to patient-oriented research. Without these highly qualified people, patient-oriented research centres cannot offer the necessary support to the local community of health care professionals and researchers, assist in developing local themes of excellence, and participate in nationwide research efforts. Nor can they coordinate local activities and recruit additional patients in clinical studies.
2.3.3 Financial Constraints
Public sector investments
By its latest calculations, CIHR, the largest public funder in Canada of patient-oriented research, invests approximately $115 million per year, or about 12% of its current budget, in funding for patient-oriented research through its various open and strategic grants and awards programs. This figure excludes financial support to various networks, platforms and knowledge translation activities categorized as patient-oriented. Public investments in patient-oriented research in Canada also include provincial government investments as well as donor-funded investments through Canada's national health charities.
Public sector investments in Canada for large multi-site clinical studies are limited at the present time. A key reason is that these studies have become much more expensive due to additional rules governing research and increased administrative requirements and, in some cases, because of their international nature.46 Yet, there is a growing need to conduct intervention studies and trials that are large enough to address important clinical issues and undertake observational research projects that can identify health gaps across many centres. The results from such studies provide the basis for firm recommendations about patient diagnosis, prognosis and treatment. Because of the ever-increasing costs and limited public funds, prospective investigators must identify a combination of potential private sector partners, public agencies and charities to assemble the required funds for major projects. This process can take several years, even for potentially practice-changing research.
This tight funding situation has been compounded recently by a decrease in indirect financial support from the university and hospital sectors. University departments traditionally have supported salaries and protected time for research, while hospitals have allowed researchers the use of clinical laboratories and diagnostic tests. However, because of budgetary constraints in all provinces and territories, institutions have moved to a cost-recovery approach. Thus, equipment, facilities and personnel are no longer available to clinical researchers without funding to reimburse the institutions, thereby reducing the capacity to conduct patient-oriented research.
Private sector investments
Canada has a strong track record of partnerships between academia and the pharmaceutical industry. The top 10 pharmaceutical companies spent more than $1.3 billion in research and development in Canada in 2007. Of this amount, $600 million were spent on clinical trials in Canada, accounting for 78% of total applied research expenditures.47 However, in Canada, as in other Western countries, processes for enrolling patients and performing trials are considered slow and costly, and have led to a shift to conducting clinical trials in developing countries.46 As a result, investments in clinical research and the overall investment in research and development in Canada by the pharmaceutical industry have flattened over the last five years despite overall global growth48 (Figure 6).
Canada's position as a world leader in clinical research is threatened by serious competition from Eastern Europe, India, China, Brazil and Russia. These countries provide rapid access to large numbers of patients, while conducting research there costs 30% less than in Western nations. The potential to gain access to emerging markets is also quite attractive to industry. China's market, for example, is growing at a rate of 25% per year. The lack of intellectual property protection is no longer a barrier to conducting research in many developing countries. In addition, China and India are investing billions of dollars to provide the infrastructure, capabilities and training necessary to run clinical trials and attract industries. Canada's share of global clinical trials has consequently shrunk to 4.1%, representing an average relative annual growth decline of 12% and placing our country fourth after France, with 4.3%, and Germany, at 5.7%. The United States leads, with 49% of global trials.49 Notwithstanding this shift, recent evidence suggests that the quality of randomized controlled trials (RCTs) conducted in developed countries such as Canada remains superior to the quality of RCTs conducted in China and India which are not meeting international standards for quality.50-52
The growth of multinational pharmaceutical companies' in-house research and development investments in Canada has also slowed since 1999 and is proportionately lower than in competitor countries, despite higher revenues. While the 2005 research and development investment by multinational pharmaceutical companies was at 19% of sales in the United States and 25% in the United Kingdom, it was only at 9% in Canada.
Figure 6: Number of Clinical Trials between 2003 and 2007 in Canada Led or Sponsored by the Pharmaceutical Industry
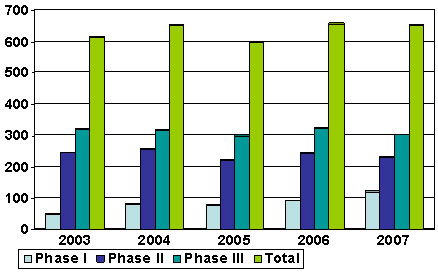
Figure 6 depicts the lack of growth in the number of industry-led clinical trials and equivalent lack of growth in investment in research in Canada over the years 2003 to 2007. Data suggest that numbers have not changed significantly for Phases I, II or III since 2003.
When therapies are developed and tested in Canada, it ensures that Canadians are the first to access new drugs and technologies. It provides opportunities to train our health care professionals in the use of these drugs and therapies, assists in their dissemination and generates economic benefits. Should the studies fail to show benefit, Canada is then in the best position to make decisions about limiting the use of those drugs and procedures. These investments and partnerships also contribute to the development of a culture of enquiry by constantly challenging existing approaches to patient care. Consequently, they foster a more knowledge-based workforce.
2.3.4 Lack of Standardized Data and Technology Platforms
Lack of standardized data and technology platforms that support a broad range of health research across institutions, research teams and provinces is an important infrastructure concern. Technology platforms allow the sharing of project-based information within networks, the coordination of research activities and the implementation of common standards, as well as easier measurement of outcomes. In addition, these electronic platforms can link to electronic medical records. While more observational, administrative and clinical databases are being put into place in parts of Canada, there are a number of barriers continuing to confront researchers in the use of this data for research processes.
Data definitions and the type of data collected are often not standardized between databases, making it difficult to compare results both nationally and internationally and to measure progress over time. In addition, the process for cleaning and linking the data are time-consuming and cumbersome and data-managers and analysts are in short supply. Additionally, much of the administrative data being captured continues to be incomplete.
Electronic data capture (EDC) tools that facilitate data collection especially in clinical trials can increase data accuracy, decrease the time to collect the data and support randomization. Yet, only about 40% of clinical trials in Canada use electronic data capture tools with trials funded by academic institutions, government and foundations much less likely to use an EDC system than industry-sponsored trials.53
Finally, while welcome progress is being made with respect to the development of electronic health records, Canada remains far behind many other countries in this regard. Additionally, individual provincial privacy regulations limit the sharing of data between provinces and there are as yet no plans on the part of the Canada Health Infoway to allow the use of this valuable information for research purposes.
2.3.5 Delays in the Approval of Large, Multi-Site Clinical Studies
Six recent reports on clinical research39-44 have emphasized that Canada has significant deficits in the leadership and coordination of patient-oriented research, especially in clinical studies, despite the potential enormous impact of these different types of studies on clinical care. Figure 7 briefly outlines the types of clinical studies.
Figure 7: Types of Clinical Studies

Figure 7 outlines the types of clinical studies, highlighting that clinical studies encompass more than just clinical trials.54
With its strong reputation in clinical research, Canada has been an active player in all types of clinical studies including large observational studies that are essential building blocks for practice-changing comparative studies or clinical trials. On the whole, decisions with respect to all types of clinical studies are generally seen as sound and protect the public interest. That being said, there are a number of systemic barriers that produce significant inefficiencies and delays with respect to undertaking large clinical studies in Canada. These inefficiencies relate, in part, to some of the data issues noted as well as the human resource gaps in terms of clinical investigators, biostatisticians, clinical epidemiologists and other required expertise.
There are some particular challenges in Canada related to large, multi-centre randomized controlled trials (RCTs), which are leading to costly delays in approvals.
The first major impediment arises from the existing requirements for complex and multiple ethics reviews. At present, RCTs must undergo full ethics reviews in each institution in which a project is conducted. This may involve as many as 30-50 separate reviews and approvals leading to important delays in the conduct of the trials and the eventual lack of inclusion of some trial sites. There is currently very little evidence to suggest that these multiple reviews have resulted in improved oversight or have addressed important ethical and patient safety concerns. These multiple reviews in fact tend to limit transparency and accountability in how decisions are made as well as being very resource-intensive.
The second challenge relates to the lack of standardized contracts between institutions involved in the study, since reviews and approvals of contracts between industry, investigators and multiple institutions have become laborious and time-consuming. There is no template or model agreement accepted by institutions that would facilitate contract signing. In fact, each site in a multi-centred trial is negotiated separately. Studies have shown that it takes an average of 96 days per site to complete a clinical trial agreement in academic centres in the U.S., whereas in Canada negotiation times can be as high as twelve months per site.55 The larger the trial, the longer and more laborious the process.
2.3.6 Deficiencies in Guideline Development, Dissemination and Uptake
Clinical practice guidelines, also called clinical care guidelines, are systematically developed statements to assist health care practitioners deliver appropriate, evidence-informed care to patients. Guidelines make explicit recommendations for care with the specific intent to influence what health care providers do. That being said, guidelines are indeed guidelines, not rules; they support clinical decision-making, not supplant it.
Developing best practice guidelines is a complex, rigorous and resource-intensive process. Over time, best practices in guideline development has advanced from informal consensus development, which resulted too frequently in guidelines of poor quality, through a more formal consensus development process. Today, there tends to be a much more evidence-based guideline development process, where rules of evidence are emphasized over expert opinion. Going further, explicit guideline development builds on evidence-informed guidelines by clarifying the rationale for the recommendations, estimating the possibility of the outcomes and comparing the desirability of the outcomes based on patient preferences.56
Steps in the development and dissemination of practice guidelines are numerous. First, a regional or national body selects a topic in a clinical area where a need is identified. Panel members are then selected. Data are synthesized by systematically searching the literature, including existing guidelines and weighing the strength of the evidence. These data are then peer-reviewed, considered in the light of clinical experience and distilled into guideline recommendations. The sponsoring organization and other interested parties endorse the guidelines. The guidelines are then disseminated through mailings, publication in professional journals and other means, including their inclusion in continuing professional development sessions. Finally the guidelines are usually subjected to re-appraisal at some point and the process begins again.57
Studies consistently demonstrate that when evidence-based practice guidelines are rigorously developed and effectively implemented, both quality of care and health outcomes improve.58 The uptake of new evidence could also result in significant cost savings to the system. For example, if practice changed to reflect the current evidence with respect to the self-monitoring of blood glucose levels, more than $150 million would be freed up to be spent elsewhere and patient health would not be negatively affected.59
Yet, the evidence shows serious deficiencies in the quality of guidelines as well as their adoption into practice. For example, with the exponential growth in practice guidelines development clinicians are increasingly being confronted with differing and sometimes contradictory disease-specific guidelines. Serious concerns have also been raised about the quality of guidelines being developed. Many primary care physicians find individual, disease-specific guidelines to be frustrating in practices increasingly characterized by older patient populations with co-morbidities. Lack of ability to critically appraise the quality of clinical practice guidelines is considered another barrier to their use. One Canadian study revealed that the developers of Canadian drug therapy guidelines are producing guidelines perceived to be clinically useful to physicians and pharmacists, but which were developed by processes not deemed rigorous or thorough.60 A more current study focusing on guideline development in Canada revealed that guideline development processes have improved in some areas, but not in others. Guidelines produced more recently in Canada were less likely to be based on a review of the evidence, and only about half discuss the levels of evidence underlying recommendations.61
In Canada, guidelines are developed by a range of groups including professional associations, volunteer groups of clinicians and health charities, often with limited resources. The Canadian Stroke Guidelines, which have proved invaluable for changing stroke care, have been funded by the Canadian Stroke Network through its NCE funding, a stable source of funding that will end in 2014. A traditional source of funding for guidelines has been provided by the pharmaceutical and medical device industry, but this source has been drying up as drugs come off patent and for other reasons. Additionally, despite strict guidelines being in place for receipt of these industry funds, conflict of interest concerns remain high among patients and the general public.
The myriad guidelines and the range of guideline developers results in a duplication in searching and appraising of evidence. In some situations, lengthy gaps in updating guidelines result in guidelines not reflecting the most recent evidence-based recommendations.
There has been recent progress, however, in addressing some of the deficiencies. The Task Force on Preventive Health Care re-established by the Public Health Agency of Canada is worthy of note. It is mandated to develop clinical practice guidelines for primary and preventive care that support primary health care providers in delivering preventive care by harmonizing various prevention guidelines and developing related practice tools and resources. While the target audience is primary health care professionals, important secondary audiences include community and public professionals (public health nurses, nutritionists), program developers, policy makers and the Canadian public.
The development and harmonization of practice guidelines are only the first steps. To be effective, guidelines have to be widely and regularly disseminated to physicians and other health care practitioners in a timely manner, over a sustained period of time and in multiple formats. This, too, can be an expensive and laborious process. The Canadian Medical Association maintains the CMA Infobase, a repository or clearinghouse of Canadian clinical practice guidelines aimed at medical practitioners. However, the Infobase does not contain guidelines for professions other than medicine, leaving an important gap in terms of access to guidelines targeted to other health professionals. Finally, with the increasing focus on self-care, home-based care and the need for individuals to take greater responsibility for their own health, there is an increasing need to translate guidelines for patient use and to finding different distribution channels to reach this audience.
Once all these steps are done, the timely adoption of guidelines by practitioners in a way that optimizes health care creates a whole new set of challenges. The evidence shows serious inefficiencies in the adoption of guidelines in practice. In fact, undoubtedly the greatest challenge with respect to clinical practice guidelines is ensuring their timely adoption by practising clinicians and other health professionals.
The reasons for the poor uptake of guidelines are numerous and complex. They include:
- The quality of the guidelines;
- Characteristics of the health care professional, including demographic variables such as age and country of training;
- Characteristics of the practice setting, including system inefficiencies and implementation barriers, workload, incentives or disincentives, existence or absence of regulation by accreditation or licensing bodies; and
- Patient factors such as individual demands from patients or individual presentations and compliance patterns.62
Unfortunately, the creation of guidelines without successful uptake becomes a fruitless exercise.
2.3.7 The Limited Role of Patients in Patient-Oriented Research
The traditional role of patients and their families has been as participants in clinical studies. The CPRN study on clinical networks commissioned by CIHR found that it was rare for patients or the public to be involved in developing, analyzing or communicating the results of studies undertaken by the networks. Yet, a growing body of evidence supports the inclusion of patients and the wider public in setting national research agendas and incorporating patient and public preferences and values into health research.63 The limited role of patients is becoming of increasing concern to many. Health services research studies document a long history of advocating for more involvement of patients and the general public in deciding research priorities and agendas,64-66 and there is increasing evidence on how valuable this can be.
In some areas of health care provision, most noticeably in maternity care, there is a long history of direct involvement of patients in the questioning of practice and policy, often as a result of views that differed markedly from accepted practice at the time. The widespread use of routine episiotomy in the 1970s, which was purported by physicians to have a number of advantages over restricted use, was repeatedly questioned by women. The results of an eventual clinical trial showed no benefit from routine use.67 Similarly, active advocacy on the part of women for a clinical trial on the benefits of radical mastectomies lead to cessation of this invasive procedure when no health benefit was shown over the use of the less invasive lumpectomies.
A significant disconnect seems to continue to exist with respect to the prevalent focus of clinical trials on drugs as opposed to other forms of intervention. For example, the research priorities of patients with osteoarthritis of the knee and the clinicians looking after them favoured more rigorous evaluation of physiotherapy, surgery and assessments of coping strategies. Only 9% of patients wanted more research on drugs. Yet, over 80% of randomized controlled trials in patients with this condition were drug evaluations. The interest in non-drug interventions in users of research results is also reflected in the fact that the vast majority of the most frequently consulted Cochrane reviews concern non-drug forms of treatment.29
There is also emerging interest in patient reported outcomes, in addition to clinically reported outcomes. This is a huge emerging area of study in the United States and Europe. The concern is that clinical trials focused on particular medical endpoints and not on the patient experience with drugs and devices might result in an efficacious drug being designed that brings with it a quality of life or personal cost too great to warrant the use of the drug. An example is provided by people with AIDS, first in the United States and then in the United Kingdom, who challenged researchers' approaches to conducting trials which had overlooked patients' preferred outcomes.68
The disability community in the United States, as well as many in the aboriginal community and HIV/AIDS communities in Canada have insisted that patients and the public have a more active role in determining research priorities as well as research being more participatory and including them in the research process – the notion of 'nothing about me without me'.
Challenges exist in terms of research participants. For example, there are remarkably few women enrolled in clinical trials in Canada leading to biases in the results. As a consequence, women are at greater risk of suffering adverse drug reactions form medications already on the market because of their under-representation in clinical trials.69 Additionally, a number of drugs or devices that are eventually approved for patients may not be efficacious for use in women. Low dose aspirin, for example, while effective in reducing risk of myocardial infarction in men, subsequently proved to be ineffective in women.
Even when women are included in trials, the data is not necessarily analyzed by gender. A systematic review of the incorporation of sex and gender into research designs has found that, while there has been a progressive improvement since the 1990s, there still exists a striking under-representation of research about gender differences in patient care.70 In fact, the dosing of most drugs is not corrected for body weight. For drugs with narrow therapeutic ranges or steep dose concentration curves, this may cause significantly increased adverse events in females compared to males.
3.0 The International Landscape
Innovation and investments in patient-oriented research have become a high priority in the United States,71 the United Kingdom,72 Europe (Germany, France, Scandinavia, Italy),73,74 and Australia.75,76 Recognizing the importance of clinical research in securing their national health and international competitiveness, these countries have invested massively in rebuilding their clinical research enterprises to better capture the health benefits and economic potential of this important sector.
The United States
In 2002, the National Institutes of Health's (NIH) Roadmap identified the need to re-engineer the United States (US) clinical research enterprise. In fiscal 2007, total Roadmap funding was $483 million. It was increased to $496 million in fiscal 2008, and grew to $534 million (as part of the overall $9 billion NIH investments for clinical trials and other patient-oriented research) in fiscal 2009. In addition to Roadmap funding, the NIH invested a further $452.4 million in 2009 through two programs administered by the National Center for Research Resources: the General Clinical Research Centers and the Clinical and Translational Science Awards. The Science Awards Program assists academic institutions in integrating and consolidating systems so as to advance and nurture a cadre of well-trained multi- and transdisciplinary researchers and research teams, create an incubator for innovative research tools and information technologies, and catalyze the application of new knowledge to clinical practice. Each Center receives $60 million per year to implement this mandate.
The United States' clinical research enterprise has also benefited from investments of $1 billion from the Department of Veterans' Affairs and $330 million through the Agency for Healthcare Research and Quality, $79 million of which was earmarked for patient safety research. This is in addition to the previously-mentioned Recovery Act of 2009 investment of $1.1 billion for clinical effectiveness research. (Note: these figures are in US dollars.) The Obama administration has recognized the importance of research to underpin system sustainability. It recently invested in comparative evaluations as a means of improving care and minimizing costs in the US health care system arguing that savings from research will offset the costs of health reform.77
The United Kingdom
The United Kingdom (UK) has made a series of impressive strides in patient-oriented research. As an early step, large comparative studies were developed to inform health decisions for the National Health Service (NHS). This premise is, in large part, the basis for the mandate of the UK National Institute for Health Research,78 were 53% of investment in research and development is targeted specifically at the infrastructure for clinical studies and systems research.79
In 2003, the UK began reorganizing its clinical research infrastructure to improve efficiency and break down barriers. At that time, 300 independent National Health Service (NHS) trusts operated independently. This fragmentation made it extremely difficult to conduct multi-centre trials. Individual decision-making authority, combined with unreliable recruitment, variations in data management and inconsistent training procedures caused delays in study timelines and drove up costs. Based on the recommendations from several reports, including Research for Patient Benefit80 and a review of UK health research funding,81 the government implemented two national initiatives to overcome these obstacles: the UK Clinical Research Collaboration and what is now known as the National Institute of Health Research (NIHR) Clinical Research Network.
The UK Clinical Research Collaboration is a partnership among the National Health Service, public and private research funders, industry, regulatory bodies, Royal colleges, patients groups and academics. It oversees patient-oriented research at the national level. The NIHR Clinical Research Network coordinates eight research networks in England, working in close collaboration with similar programs in Scotland, Wales and Ireland. Collectively, these networks cover six topic areas (neurodegenerative diseases, mental health, stroke, cancer, diabetes and medicines for children); primary care; and coordinating centres (which provide comprehensive support for the six topic networks and the primary care network).
The UK has also moved forward in developing common research contracts for multi-site clinical trials with the pharmaceutical industry's input and involvement and to ensure that research priorities would be aligned and responsive to industry's needs.
The NIHR's Collaborations for Leadership in Applied Health Research and Care (CLAHRCs), established in 2008, was a response to the identified need for the National Health Service to better harness the capacity of higher education to support initiatives to enhance the effectiveness and efficiency of clinical care. Each of the nine collaborative partnerships between a university and its surrounding NHS organizations will receive up to £10M a year over five years plus matching funding to focus on the "second gap in translation" – translating research into practice.
In another bold step, the UK established the National Institute for Health and Clinical Excellence (NICE) in 1999. NICE's mission is to promote clinical excellence and efficient use of health system resources. The organization is responsible for establishing, maintaining and promoting clinical standards for the UK health system, providing guidance, and maintaining a national database. NICE also paved the way for greater involvement of patients and caregivers ranging from involvement in setting priority topic areas for clinical guideline development, membership in guideline development groups, and as members of research teams contributing to the development, implementation and evaluation of research commissioned across its funding programs. The Standing Advisory Group for Consumer Involvement in R&D (INVOLVE) develops strategic alliances among key groups to maximize effective public involvement and empower individuals to play an active role in research.
4.0 The Strategy
4.1 Vision
By 2025, Canada will have demonstrably improved health outcomes and enhanced the health care experience for patients through the integration of evidence at all levels of the health care system.
4.2 Goals
- To grow Canada's capacity to attract, train and mentor health care professionals and health researchers in patient-oriented research and create sustainable, patient-oriented career paths across the breadth of health disciplines.
- To establish an integrated, leading-edge, pan-Canadian clinical research infrastructure along the full continuum of patient-oriented research.
- To strengthen organizational, regulatory and financial support for clinical studies in Canada and enhance patient and clinician engagement in these studies.
- To improve processes for the early identification of best practices, expedite their development and harmonization into guidelines for patient care, and support their adoption by clinicians, caregivers and patients.
- To create a collaborative, pan-Canadian process for identifying, establishing and addressing patient-oriented research priorities.
4.3 Principles
The Strategy is built upon the following principles:
- Public Interest – Ensuring that the public interest is of paramount importance in the creation and use of knowledge supported by the strategy.
- Collaboration – Working in common cause with partners and key stakeholders on the development and implementation of the Strategy and on achieving its goals.
- Responsiveness – Ensuring that identified areas of greatest need drive the research and knowledge translation agenda.
- Excellence – Expanding on existing clusters of excellence and developing excellence where it does not currently exist.
- Inclusivity – Encompassing the breadth of clinical settings, health professions/disciplines and constituent voices across Canada.
- Innovation – Fostering a culture of constant questioning and a commitment to new ideas and novel solutions.
- Accountability – Applying the highest ethical and financial standards, ensuring that the appropriate metrics and evaluation processes are in place to evaluate the Strategy's impact and the attainment of its goals.
- Transparency: Making decisions openly and engaging partners and citizens in the discussion.
4.4 Bridging the Research-to-Practice Valleys
A series of initiatives are proposed to bridge the research-to-practice valleys and to move Canada to a position of leadership in patient-oriented research. The initiatives are less discrete elements than they are parts of a whole, wherein one element lays the groundwork for another and successive implementation creates the capacities, synergies and trust necessary to meet the goals of the Strategy.
Some of the recommended initiatives are narrower in scope and have the potential for some early wins given an existing degree of consensus on the nature of the problem and some early work having been completed (i.e. addressing the concerns around ethics reviews and standardized contracts for large multi-site clinical trials). Others, such as developing the clinical research talent pool, are long-term initiatives. Yet, there is an imperative to begin immediately given the breadth of the challenge, the need for engagement of many partners and the timeframe in which progress can be realized. The Patient-Oriented Research Networks initiative has both immediate and longer term recommendations. Improved processes for the development and dissemination of clinical practice guidelines need to begin immediately, requiring more research to better understand the factors affecting uptake so as to put in place the most effective strategies to address this challenge.
Strategies to involve patients in a meaningful way in patient-oriented research are imbedded in each of the recommended initiatives, as patient and public engagement is integral to the Strategy as a whole.
4.4.1 Establishing Collective Leadership
Collective guidance and leadership for the Strategy from both levels of government is essential to meeting the Strategy's goals. Both levels of government are integral players in each component of the Strategy, with the health care system and economic benefits largely accruing to the provinces. While progress can be made on the Strategy's goals without the full engagement of all provincial/territorial governments the kind of integrative, transformative change that is envisioned and required will not be realized without shared governance of the Strategy and collective engagement and leadership. This, however, will undoubtedly take time. A focused round of consultations on this document with provincial/territorial governments is planned as an initial early step.
There is also opportunity and a will to move forward quickly on a number of fronts. A strong commitment exists from a cadre of government and non-governmental agencies as well as national champions from the voluntary sector and industry to facilitate progress on a number of the Strategy's specific elements and create momentum for the Strategy. CIHR has demonstrated its strong commitment and readiness to play a leadership role and a number of exciting initiatives are already underway.
The first step forward is the establishment of a national Steering Committee with members drawn from the federal and provincial/territorial governments, health research funders, health researchers, health information and assessment agencies, academic health centres and universities, relevant industries, professional associations and the patient community.
It is proposed that the Steering Committee be chaired by the President of CIHR with a co-chair or vice-chair from outside the federal government. The Steering Committee will be guided by the principles of the Strategy and its members committed to driving towards the Strategy's vision. Its secretariat, housed in CIHR offices, would be led by a national executive director. Much of the work of the Steering Committee would be undertaken in expert working groups with clear mandates and accountabilities and bringing specific expertise and a range of perspectives to the table.
An early task for the Steering Committee will be the development of an implementation plan with concrete timelines, deliverables and evaluation metrics as well as an initial costing of the Strategy's elements. The creation of a pan-Canadian process to establish patient-oriented research priorities and the broad themes of greatest importance to Canadian health outcomes is also seen as requiring early Steering Committee attention. Fully engaging CIHR's Scientific Directors as well as the health charities, the provincial health research funding bodies, patients and the public in this process will be of critical importance.
4.4.2 Developing the Talent Pool
For clinical investigators to thrive, a supportive environment is essential.33-38
All clinical investigators, independent of their domain of expertise, are critical to patient-oriented research and require better support. Clinical investigators play roles in training the next generation of researchers, but themselves require access to better training and support structures. Clinical investigators are also pivotal to asking the right questions and knowing how to answer them, yet often cannot compete in the current environment for career awards. They also contribute to the timely knowledge translation across the continuum from discovery to application to policy. Canada needs treatments that are evidence-informed and clinical investigators are ultimately those that gather the evidence. While some clinical investigators can and do compete successfully for national career awards, there are simply not enough investigators. Furthermore, there are serious shortages of clinical investigators focusing their research programs on clinical and health services issues.
Therefore, to build capacity in patient-oriented research, Canada must re-engineer career training and salary awards for clinical investigators. Awards will be designed to:
- consider different practice plans and remuneration strategies (e.g. alternate payment plans and fee-for-service for physicians), types of positions (e.g. university-based), and sources of support for other health science professions;
- take into account the environment (e.g. the institutional commitment to the applicant and the program in which they work) as well as the individual's achievements; and
- go beyond providing salary support to include recruitment of team members and program development, as well as infrastructure.
CIHR is well-positioned to move ahead and play a leadership role in this area.
Canada needs more methodologists to support patient-oriented research and more health care professionals, especially those with advanced degrees and trained in the core methods necessary to carry out research activities. This will require universities nationwide to better support master's and doctoral programs in areas such as epidemiology and biostatistics, health economics and other health-related social sciences. It also implies developing standardized training for research coordinators, project managers and other support personnel. Identifying and liaising with the entities responsible for training research personnel will be essential in order to address the full career spectrum in a coordinated fashion. Discussions on how best to move forward will need to include not just universities, but perhaps also provincial ministries of education and professional associations.
Individual awards should require contributions from clinical or academic departments, hospitals, universities and regional or local health authorities as appropriate. They should be linked to regular monitoring of the recipient's performance and the institution's commitment to the researcher.
4.4.3 Enhancing the Conduct, Funding and Effectiveness of Multi-Centre Clinical Trials
All large clinical studies are best managed using standardized policies, procedures and templates that, as much as possible, incorporate international best practice while responding to unique Canadian needs. There is an emerging opportunity to move forward initially and quickly to resolve the specific barriers to the conduct of large, randomized controlled trials in Canada. Progress made with respect to RCTs can then be adapted to meet the needs of other kinds of clinical studies.
In terms of ethics reviews, the recent experience and advances in the United Kingdom, Australia and France, where common ethics reviews have been adopted, provide important models for Canada to consider. In addition, British Columbia, Alberta, Quebec, Newfoundland and Labrador, and Cancer Care Ontario have all developed, or are moving to, models of single reviews which provide a strong basis for moving forward.
In terms of standardized contracts, the aim is to develop templates which can be agreed to by every province and territory with the recognition that, once developed, they will require adaptation to accommodate local laws and government regulation. Again, there are models and recent experiences to draw from. A model clinical trial agreement has been adopted at the national level in the United Kingdom by all relevant stakeholders. Other countries, including Australia, the United States and Germany, are at various stages of development of such agreements although none have yet received full acceptance. In Canada, there has been some recent progress in a number of provinces. Guidance documents for negotiating clinical trial agreements have been developed in Ontario, spearheaded by the Ontario Institute for Cancer Research. In Quebec, a proposed clinical trial agreement generated within the Ministry of Health is being discussed, and there has been some progress made in Alberta by the Northern Alberta Clinical Trials Research Centre. At this time, there is interest among both the Canadian pharmaceutical industry and the academic health care organizations in developing a pan-Canadian model clinical trial agreement. It would be intended for use between industry sponsors and academic health care centres, particularly for multi-site clinical trials.
Once developed, the proposed Patient-Oriented Research networks and SUPPORT units described below can be used as vehicles for the implementation of these standard agreements and can play a role in ensuring that all interventions designed to improve timeliness and efficiency are benchmarked, evaluated and reported on to ensure that targets and objectives are being met.
At present, there is not enough funding available to support clinical trials that are desperately needed to begin to answer important health care related questions for Canadians. Developing a set of needs-based priorities for clinical trials in Canada with clear patient involvement in establishing these priorities could also facilitate funding from public sources such as the different levels of government and Canada's health charities and non-profit foundations. In particular, our country must make increasing investments in publicly-funded clinical trials if we are to regain lost ground and move forward to a position of world leadership in patient-oriented research. Partnerships among academia, industry and health charities are essential to enhance funding for important patient-oriented research in Canada.
Success in establishing agreements on ethics reviews and standard contracts should lead to increases in private funding of large multi-site projects which often require a patchwork of funding sources. This will be particularly true if key funding partners are part of the process of developing the templates and standardized agreements. Reaching agreement on these issues will also increase Canada's competitive advantage by making it more appealing to international companies wanting to efficiently and cost-effectively conduct clinical trials and other large clinical studies in a timely fashion.
4.4.4 Focusing on Patient-Oriented Research Networks
Patient-oriented research networks
As noted, patient-oriented researchers in Canada already have impressive experience in working together in multidisciplinary or transdisciplinary networks or on expert teams. However, strategies are required to enhance the sustainability of their networks, link them to each other and provide them with the methodological support needed to meet their research goals.
As part of the Strategy, CIHR's Scientific Directors and representatives of the various funding partners and stakeholders will identify key clinical research areas for the creation of patient-oriented research networks. These research networks will identify research priorities and define clinical protocols, assist in the scientific oversight of research programs, support scientists through various teaching and mentorship activities, and foster interdisciplinary research. They will provide the scientific leadership necessary to direct focused research and specific projects most relevant to Canadians. They will assist in patient recruitment strategies for large multi-centre studies and oversee the conduct of those trials. They will be primarily solution-focused, and will link existing and emerging researchers or centres of excellence across the country to undertake patient-oriented research in designated health areas. They will focus on outcomes-oriented research that meets the needs of relevant stakeholders and receptors of the research: clinicians, industry, policy makers, patients and the public. They will need to demonstrate access to the necessary methodological expertise and research support and provide a plan for building capacity in both areas. Each network will be expected to demonstrate how it will ensure project specific support (e.g. project management, methodological assistance, data management and statistical services).
To achieve these objectives, each patient-oriented research network will have the resources to ensure leaders have protected time, administrative support and sufficient funding. Networks will be expected to:
- create a critical mass of multidisciplinary scientific expertise;
- link to other researchers in their area through regular meetings and planned communications;
- develop a coordinated, nationwide patient-oriented research agenda;
- develop a coordinated funding approach vis-à-vis relevant funders;
- Integrate SUPPORT unit(s) (see below) that will provide methodological, biostatistical and specific project management assistance;
- assist in conducting research and disseminating results from academic settings into the wider patient care community;
- provide education and mentorship to emerging researchers in their field;
- coordinate overall project-specific research activities to support their theme;
- ensure maximum efficiency in enrolment in large-scale trials; and
- ensure that each large-scale study meets the highest ethical standards and conforms to national operating standards.
The Drug Safety and Effectiveness Network (DSEN), which is complementary to this Strategy, is an example of a patient-oriented research network that was created to focus on a particular theme, identified as a priority by both the provincial and federal governments (consumer safety). It is intended to focus largely on observational research, particularly pharmacoepidemiological studies, and may also venture into clinical trials to accomplish its mission. While DSEN was developed prior to this Strategy, it is an excellent example of a thematic research network.
With respect to the networks, it is anticipated that there will be significant interest in partnering among the public sector health charities, as the networks will focus on specific topics that are consistent with many of the charities' mandates.
4.4.5 Building and Resourcing SUPPORT Units
Patient-oriented research units
Another element of the Strategy will be the creation of specialized research service centres referred to as Support for People and Patient-Oriented Research and Trials (SUPPORT) units, in an effort to provide the necessary and often highly specialized methodological expertise to those engaged in patient-oriented research.
SUPPORT units are multidisciplinary centres of methodologists and other experts who are essential to the patient-oriented research enterprise. These experts include, but are not limited to, clinical epidemiologists, biostatisticians, social scientists, health economists and clinical trialists, as well as research support staff including research coordinators, research associates and assistants, data collectors and data analysts. Their role is to assist investigators design relevant research studies (including observational and interventional studies), conduct biostatistical analyses, manage data and biobanks, provide and teach project management skills, and ensure studies meet all relevant regulatory standards.
SUPPORT Units will take two forms:
1. Geographically located units will be focused on building methodological support where it does not now exist, or where the available resources are inadequate to support the research ongoing and contemplated in a particular location. Using an integrated knowledge translation model, they will train more people, and be directly involved in local research decision making.
The objectives of these SUPPORT Units would be three-fold:
- to provide needed methodological and logistical support to local clinical investigators to improve the quality of research proposals and research they undertake and to make them more competitive at obtaining research funding.
- to build the capacity of research methodologists and research support staff by providing a training ground and experience in these disciplines.
- to support decision making within the clinical setting and catchment area by clinicians, managers and administrators and implementation of best practices by working collaboratively to respond to the research and implementation needs identified by the clinical setting.
Hospital CEOs, Chairs of regional health authorities and local administrators will need to demonstrate commitment to SUPPORT by providing the necessary space and matching funding and by developing a plan for their sustainability. They will also need to demonstrate how they will engage with the units to promote the conduct of research to address local clinical and health services issues (including improving the uptake of best practices).
2. Units that are more national in scope and either provide support in highly specialized methods or provide methodological support to specific health disciplines in the clinical setting outside of medicine such as nursing, rehabilitation, dentistry and other areas to help them enhance their research skills and conduct more high quality research.
This type of SUPPORT unit will be encouraged to develop niches of excellence in areas such as randomized control trials, observational studies using large administrative databases, systematic reviews or knowledge translation science and guideline development, which will make them competitively attractive internationally. They may also be more virtual in nature and provide a methodological hub to support nursing, rehabilitation or other disciplinary research, thereby encouraging nursing researchers, for example, to share this resource to improve the quality and competitiveness of nursing research.
4.4.6 Improving Guideline Development, Dissemination and Uptake
A concerted, well-resourced, collective approach is needed in Canada to ensure that evidence-informed practice guidelines are rigorously developed and effectively implemented so as to improve quality of care and health outcomes. Recommended elements of such an approach are:
-
Developing and offering training courses and modules for guideline developers. Well-designed training modules would facilitate the work of guideline developers, enhance coordination among them, and encourage the most rigorous approach to the development of evidence-informed guidelines. Training would also serve to imbed in the development process a need to harmonize the guidelines with other relevant guidelines. Developers would also be expected to collaborate on tasks to expedite guideline development.
-
Supporting guideline development. Developing evidence-based guidelines is an expensive process, estimated as costing between $100,000 and $1 million to produce one set of guidelines. It is proposed that matching funds be made available from provincial and federal governments to guideline developers if they meet criteria which will be established regarding requirements for rigour of both the development process and guideline content.
-
Engaging patients in the guideline development process. Greater involvement of patients and caregivers in the guidelines development process, from involvement in setting topic area priorities to membership in development groups, is important not only for the quality of the guidelines but also for their ultimate uptake. The NICE initiative in the United Kingdom, described earlier, provides important learnings to help Canada develop a strategy in this area.
-
Increasing capacity for knowledge synthesis. While Canada has an international reputation in this area and produces excellent work, there is a need for more capacity in this area. As a first step CIHR should increase its investments in the Cochrane Centre and take the lead in exploring a collaborative partnership between the Canadian Cochrane Centre and other organizations in Canada engaged in knowledge synthesis, including CHSRF and CADTH.
-
Developing a comprehensive knowledge translation (KT) strategy. Current strategies for communication and dissemination of guidelines such as publication in major medical journals, continuing professional development programs, conferences and seminars have been shown to have had limited success in changing professional practice.82,83 Bringing about guideline adoption and sustaining guideline use is complex and challenging. Several studies have shown that adherence rates often go back to baseline levels after implementation strategies are discontinued.84 Unfortunately, the evidence suggests that guideline dissemination and implementation activities in Canada have actually decreased between 1994 and 2005, meaning that the potential positive impact on patient health outcomes is likely not being realized.61
Recommended activities include:
-
Funding more research on the barriers to uptake and strategies to speed guideline uptake. Considerably more research is needed to better understand the systemic, organizational and individual barriers to the uptake of guidelines by health systems, clinicians and patients, and to provide evidence on the most effective dissemination and implementation strategies.
-
Adapting the organization of care. Guideline implementation is often unsuccessful unless the organization of care is adjusted or redesigned to conveniently permit the use of evidence-informed best practices by those within the health care system. Provincial and territorial health systems will need to be engaged and work to find ways to help enable implementation of evidence-informed practice and policy.
5.0 Conclusion
By 2025 health outcomes in Canada will be demonstrably improved and the health care experience for patients enhanced through the integration of evidence at all levels of the health care system. This is the vision driving the development of Canada's Strategy for Patient-Oriented Research.
The value proposition for 'patient-oriented research' for the patient is to receive the right care, in the right place at the right time. For this to happen, health care professionals and policy makers need access to the latest evidence. This, in turn, requires the timely and efficient translation of research innovations from the research setting to patient care settings. It also calls for the evaluation and synthesis of existing knowledge and proper transfer of what we already know to the clinical setting. The value proposition for the provincial governments and health care administrators is a cost-effective, efficient, affordable health care system that meets the needs of patients and continuously improves the health of Canadians. The value proposition for the Government of Canada is to meet a number of key objectives of the Science and Technology Strategy, including the opportunity to enhance the commercialization of research.
Governments, industry and health charities will have to work together with the national Steering Committee to effect change in the field of patient-oriented research by finding solutions to inadequate funding and successfully navigating the complexities of program design and implementation.
Canada has made progress in patient-oriented research. But that progress has been slow, often scattered and hampered by federal/provincial, interdisciplinary and structural barriers. The compelling financial imperative in all provinces and territories, entrenched and emerging health challenges and increasing expectations from providers and patients alike underscore the need for a bold and innovative patient-oriented research strategy for Canada that provides a clear and shared sense of direction that will bridge the evidence-to-practice valleys and transform the health care landscape in Canada. This Strategy, with patients and the public clearly at its heart, seeks to provide that direction.
In short, the need is great, the potential benefits are huge and Canada is ready.
6.0 References
-
Evidence-Based Medicine Working Group. Evidence-Based Medicine: A New Approach to Teaching the Practice of Medicine. JAMA. 1992; 268(17):2420-2425.
-
Ferriman A. BMJ readers choose the "sanitary revolution" as greatest medical advance since 1840. BMJ 2007 Jan 18; 334:111.
-
Haynes AB, Weiser TG, Berry WR, Lipsitz SR, Breizat AS, Dellinger EP et al. A surgical safety checklist to reduce morbidity and mortality in a global population. N Engl J Med. 2009 March 24; 360 (5): 491-499.
-
Albani S, Prakken B. The advancement of translational medicine – from regional challenges to global solutions. Nature Medicine. 2009 September; 15 (9): 1006-1009.
-
Fang FC, Casadevall A. Lost in translation – basic science in the era of translational research. Infection and Immunity, 2009 Feb; 78(2): 563-566.
-
Wennberg JE, Gittelsohn A. Small area variations in health care delivery. Science. 1973 Dec 14; 182:1102-1108.
-
Wennberg JE. Dealing with medical practice variations: A proposal for action. Health Aff. 1984 Summer; 3(2):6-32.
-
Wennberg JE, Elliott SF, Skinner JS. Geography and the debate over Medicare. Heath Aff. 2002 Feb 13; W97-W114.
-
Fisher ES, Wennberf DE, Stukel TA, Gottlieb DJ, Lucas FL, Pinder EL. The implications of regional variations in Medicare spending. Part 1. The content, quality, and accessibility of care. Ann of Intern Med. 2003; 138:273-287.
-
Wennberg DE, Wennberg JE. Addressing variations: Is there hope for the future? Health Aff. 2003 Dec 10; W3:614-617.
-
Weinstein JN, Bronner KK, Morgan TS, Wennberg JE. Trends: Trends and geographic variation in major surgery for degenerative diseases of the hip, knee, and spine. Health Aff. Web Exclusive. 2004 Oct 7: Var81-Var89.
-
Straus SE, Ball C, Balcombe N, Sheldon J, McAlister FA. Teaching evidence-based medicine skills can change practice in a community hospital. JGIM 2005 April: 20(4):340-343.
-
Lloyd K, Cella M, Tanenblatt M, Coden A. Analysis of clinical uncertainties by health care professionals and patients: an example from mental health. West J Med. 2000 May; 172(5):315-319.
-
Kiesler DJ, Auerbach SM. Optimal matches of patient preferences for information, decision-making and interpersonal behaviour: Evidence, models and interventions. Patient Educ Couns 2006; 61:319-341.
-
McGlynn EA, et al.The quality of health care delivered to adults in the United States. New Engl J Med 2003; 348(26):2635–2645.
-
DuBois A, Rochon J, Lamparter C, Pfisterer J. Pattern of care and impact of participation in clinical studies on the outcome in ovarian cancer. Int J Gynecol Cancer. 2005; 15:183-191.
-
Majumber SR, Roe MT, Peterson ED, Chen AY, Gibler WB, Armstrong PW. Better outcomes for patients treated at hospitals that participate in clinical trials. Arch Intern Med. 2008; 168(6):657-662.
-
Weinsaft JW, O'Rourke MF, Nichols WW, Sharma AM, Pischon T, Engeli S, et al. Effect of Ramipril on cardiovascular events in high-risk patients. N Engl J Med 2000; 343:64-66, Jul 6, 2000.
-
Fergusson D, Glass KC, Hutton B, Shapiro S. Randomized controlled trials of aprotinin in cardiac surgery: could clinical equipoise have stopped the bleeding? Clinical Trials. 2005; 2:218-232.
-
Hébert PC. Transfusion requirements in critical care. Vox Sang. 2002 Aug; 83 Suppl 1:387-396.
-
Kwan J, Sandercock PAG. In-hospital care pathways for stroke. Cochrane Database Systc Rev. 2004; 4. Art. No.: CD002924. DOI: 10.1002/14651858.CD002924.pub2.
-
Bailey RC, Moses S, Parker CB, Agot K, Maclean I, Krieger JN, Williams CF, Campbell RT, Ndinya-Achola JO. Male circumcision for HIV prevention in young men in Kisumu, Kenya: a randomised controlled trial. Lancet. 2007 Feb 24:369(9562):643-656.
-
Murphy KE, Hannah ME, Willan AR, Hewson SA, Ohlsson A, Kelly EN, et al. Multiple courses of antenatal corticosteroids for preterm birth (MACS): a randomised controlled trial. Lancet. 2008 Dec 20/27;372:2143-2151.
-
Stevens B, Yamada J, Ohlsson A. The efficacy of sucrose to reduce procedural pain in neonates as assessed by physiologic and behavioural outcomes. Neonatal Modules of the Cochrane Database of Systematic Reviews. The Cochrane Collaboration. Eds. Sinclair JC, Brachen MB, Soll RS. Update Software, Oxford. 2002.
-
Pisters KM, Evans WK, Azzoli CG, Kris MG, Smith CA, Desch CE, et al. Cancer Care Ontario and American Society of Clinical Oncology adjuvant chemotherapy and adjuvant radiation therapy for stages I-IIIA resectable non small-cell lung cancer guideline. J Clin Oncol. 2007 Dec 1; 25(34):5506-5518.
-
Ellard SL, Clemons M, Gelmon KA, Norris B, Kennecke H, Chia S, et al. Randomized Phase II study comparing two schedules of Everolimus in patients with recurrent/metastatic breast Cancer: NCIC Clinical Trials Group IND.163. JCO Sep 20 2009:4536-4541.
-
Anis A, Nosyk B, Sun H, Guf DP, Banasback N, Li X, et al. Quality of life of patients with advanced HIV/AIDS: measuring the impact of both AIDS-defining events and non-AIDS serious adverse events. J Acquir Immune Defic Syndr 2009 August; 51(5):631-639.
-
Douketis J, Cook D, Meade M, Guyatt G, Geerts W, Skrobik Y, et al. Prophylaxis against deep vein thrombosis in critically ill patients with severe renal insufficiency with the low-molecular-weight heparin dalteparin: an assessment of safety and pharmacodynamics: the DIRECT study. Arch Intern Med 2008 Sept 8; 168(16):1805-1812.
-
Chalmers I, Glasziou P. Avoidable waste in the production and reporting of research evidence. Lancet 2009 July 4; 374:86-89.
-
Picard A. Real patient care reform could be a mouse-click away. The Globe and Mail 2010 Nov.11.
-
Nason E, Brisbois R. Clinical research networks in Canada: an environmental scan. Canadian Policy Research Networks, Inc. Ottawa. 2009.
-
Lawlor MS. Biotechnology and Government Funding: Economic Motivation and Policy Models. Science and Cents: Exploring the Economics of Biotechnology Conference. (Ed. JV Duka and MK Yücel). Federal Reserve Bank of Dallas. 2002 April 19; 132-146.
-
Frontera WR, Fuhrer MJ, Jette AM, Chan L, Cooper RA, Duncan PW, et al. Rehabilitation medicine summit: building research capacity. Executive Summary. J Neuroeng Rehabil [serial on the internet] 2006 [cited 2008 Dec 12] 3:1.
-
DeHaven MJ, Wilson GR, O'Connor-Kettlestrings, P. Creating a research culture: What we can learn from residencies that are successful in research. Educational Research and Methods.1998; 30(7):501-507.
-
Kekki P. Promoting clinical research in general practice. Educ Health (Abingdon). 2005; 18(2):283-289.
-
Cooke J. A framework to evaluate research capacity building in health care. BMC Family Practice [serial online]. 2005 [cited 2008 Dec 12] 6:44.
-
Vratny A, Shriver D. A conceptual model for growing evidence-based practice. Nurs Adm Q. 2007; 31(2):162-170.
-
Araki Y, Scott SE, Lear S. Challenges of telehealth research in clinical settings. J Telemed Telecare. 2007; 13:425-426.
-
Collet JP, Fraser W, Johnston C, Lacroix J, Paradis G, Valiquette L et al. Clinical research report. Document submitted to the Board of Directors of the Fonds de la recherche en santé du Québec. 2005 March 11.
-
Brimacombe GG, Gruenwoldt EC. Moving at the speed of discovery [electronic resource]: From bench to bedside to business. 2007.
-
Prud'homme Généreux A, Cairns J. Summary report: CIHR/CFI Clinical Research Centres/Networks Workshop. Canadian Institutes of Health Research, January 21, 2005.
-
Rouleau J. Clinical research in Canada: A proposed action plan. 2008.
-
Silverman M. A roadmap to ensure Canadian leadership in clinical and translational research. Canadian Institutes of Health Research Clinical Research Initiative. 2004.
-
Syntegrity Group. Canadian Institutes of Health Research: Clinical research syntegration – July 2008. Version 1.0.
-
National Physician Survey. National physician survey 2007.
-
SHI Consulting. Clinical trial coordinating centre business case. Report for the CT CTNet Working Group. 2008.
-
Patented Medicine Prices Review Board. Annual Report. 2007.
-
IMD. World competitiveness yearbook, 2005.
-
Thiers FA, Sinskey AJ, Berndt ER. Trends in the globalization of clinical trials. Nature Reviews Drug Discovery 7, 13-14 (January 2008) doi:10.1038/nrd2441 News Analysis.
-
Wu T, Youping L, Zhaoxiang B, Liu G, Moher D. Randomized trials published in some Chinese journals: How many are randomized? Trials 2009; 10:46.
-
Tharyan P, Premkumar TS, Mathew V, Barnabas JP, Raj M. Editorial policy and the reporting of randomized controlled trials: A survey of instructions for authors and assessment of trial reports in Indian medical journals (2004-05). Natl MedJ I 2008; 21(2):62-68.
-
Zhang D, Yin P, Freemantle N, Jordan R, Zhong N, Cheng KK. An assessment of the quality of randomized controlled trials conducted in China. Trials 2008 Apr 24; 9:22.
-
El Emam K, Jonker E, Sampson M, Krleza-Jerić K, Neisa A. The use of electronic data capture tools in clinical trials: Web-survey of 259 Canadian trials. J Med Internet Res 2009 Mar 9;11(1):e8.
-
Woodbury MG. Research 101: Developing critical evaluation skills. Wound Care Canada 2003;2(2):32-38.
-
Moldofsky M, Arts K, Evans L. Harmonization of clinical trial agreements. CAURA National Conference 2007 May 7.
-
Woolf SH. Practice guidelines, a new reality in medicine. II. Methods of developing guidelines. Arch Intern Med 1992 May;152(5):946-952.
-
Davis D, Goldman J, Palda VA. Handbook on clinical practice guidelines. Canadian Medical Association 2007. Ottawa.
-
Grimshaw JM, Thomas RE, MacLennan G, Fraser C, Ramsay CR, Vale L, et al. Effectiveness and efficiency of guideline dissemination and implementation strategies. Intl J of Technology Assessment in Healthcare. 2005; 21(1):149.
-
Canadian Agency for Drugs and Technologies in Health. Optimal Therapy Report. COMPUS, 2009; 3(4).
-
Graham ID, Beardall S, Carter AO, Glennie J, Hébert PC, Tetroe JM, et al. What is the quality of drug therapy clinical practice guidelines in Canada? CMAJ 2001;165(2):157-163.
-
Kryworuchko J, Stacey D, Bai N, Graham ID. Twelve years of clinical practice guideline development, dissemination and evaluation in Canada (1994 to 2005). Kryworuchko J, Stacey D, Bai N, Graham ID. Implement Sci. 2009 Aug 5;4:49.
-
Davis DA, Taylor-Vaisey A. Translating guidelines into practice: A systematic review of theoretic concepts, practical experience and research evidence in the adoption of clinical practice guidelines. Can Med Assoc J 1997 Aug 15; 157(4):408-416, CMA, 1997.
-
Oliver S, Armes DG, Gyte G. Public involvement in setting a national research agenda: a mixed methods evaluation. 2009 July; 2(3):179-190.
-
Chalmers I. The perinatal research agenda: whose priorities? Birth 1991;18:137-145.
-
Chalmers I. What do I want from health research and researchers when I am a patient? MBJ 1995; 310:1315-1318.
-
Oliver S. How can health service users contribute to the NHS research and development programme? BMJ 1995; 310:1318-1320.
-
Sleep J, Grant A, Garcia J. et al. West Berkshire perineal management trial. Br Med J (Clin Res Ed). 1984; 289(6445):587.
-
Thornton H. Patient and public involvement in clinical trials. BMJ 2008 24 Apr; 336(7650):903-904.
-
Fish, EN. The X-files in immunity: sex-based differences predispose immune responses. Nature Reviews Immunology 2008 Sept; 8:737-744.
-
Berlin JA, Ellenberg SS. Women in clinical trials. BMC Medicine 2009, 7:56.
-
National Center for Research Resources. Strategic plan 2009-2013. NIH Publication No. 08-6439. National Center for Research Resources, National Institutes of Health. 2008.
-
United Kingdom Clinical Research Collaboration. UK Clinical Research Collaboration progress report, 2006-2008. 2008 (United Kingdom).
-
European Strategy Forum on Research Infrastructures. European Roadmap for Research Infrastructures: Roadmap 2006. Luxembourg: Office for Official Publications of the European Communities. 2006.
-
European Strategy Forum on Research Infrastructures. European roadmap for research infrastructures: Roadmap 2008. Luxembourg: Office for Official Publications of the European Communities. 2008.
-
Department of Innovation, Industry, Science and Research, Australian Government. Strategic roadmap for Australian research infrastructure. 2008.
-
National Health and Medical Research Council. Clinical research in Australia. Report of the NHMRC Clinical Research Working Group. 2001 (Australia).
-
McKinnon JD, Bendavid N. Congress is buying time for health care savings. The Wall Street Journal. 2009 March 28.
-
National Institute for Health Research. Best research for best health. 2007.
-
National Health Service. Delivering health research. National Institute of Health Research progress report 2008-09.
-
Research for Patient Benefit Working Party. Final Report. Department of Health, United Kingdom. 2004.
-
Cooksey D. A review of UK health research funding. 2006 (United Kingdom).
-
Grimshaw JM, Thomas RE, MacLennan G, Fraser C, Ramsay CR, Vale L et al. Effectiveness and efficiency of guideline dissemination and implementation strategies. Health Technol Assess 2004; 8:1-84.
-
Grimshaw J, Eccles M, Thomas R, MacLennan G, Ramsay C, Fraser C, Vale L. Toward evidence-based quality improvement. Evidence (and its limitations) of the effectiveness of guideline dissemination and implementation strategies 1966-1998. JGen Intern Med 2006; 21:14-29.
-
Bauer MS. A review of quantitative studies of adherence to mental health clinical practice guidelines: Harvard Rev of Psych 2002 May;10(3):138-153.
7.0 Annex 1: President's Advisory Board Members – Strategy for Patient-Oriented Research
Title while on the Board
Dr. Aubrey Tingle
Former head of the Michael Smith Foundation
Dr. Arthur Slutsky
Vice-President (Research)
St. Michael's Hospital
Dr. Catriona McMahon
Vice President, Medical Affairs
AstraZeneca Canada Inc.
Dr. Jean L. Rouleau
Dean, Faculty of Medicine
University of Montreal
Ms. Sally Brown
Executive Director
Heart and Stroke Foundation of Canada
Ms. Krista Connell
Chief Executive Officer
Nova Scotia Health Research Foundation
Dr. Roger Palmer
Interim Dean, School of Public Health
University of Alberta
Dr. Brian Postl
Chief Executive Officer
Winnipeg Regional Health Authority
Ms. Susan Zimmerman
Executive Director
Secretariat on Research Ethics
Professor Dorothy Pringle
Faculty of Nursing
University of Toronto
Dr. Sarah Prichard
Vice President, Global medical affairs
Baxter International Inc.
- Date modified: