Team Grants: Lung Health
Figure 1: Research Team Composition
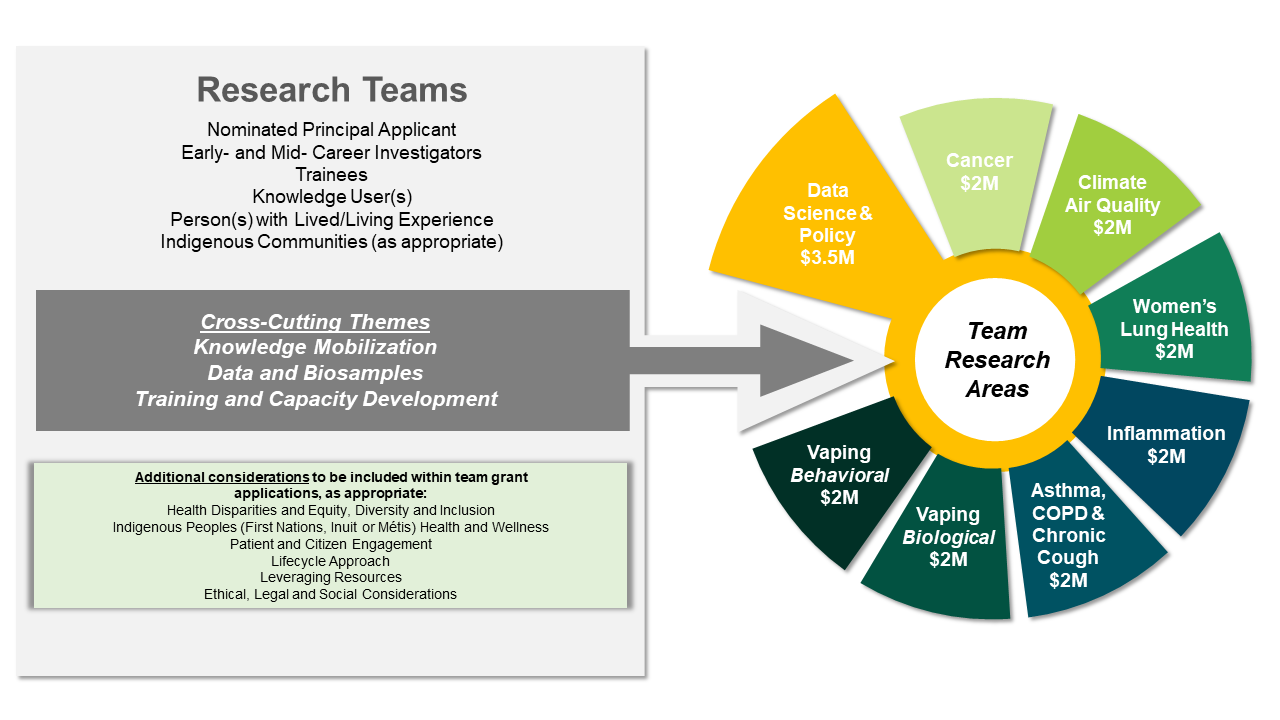
Long Description
The Team Grants: Lung Health funding opportunity includes up to eight (8) Research Teams within a defined research area. The funded Data Science and Policy Research Team will be awarded additional funds to support infrastructure and coordination costs of the Data and Biosample cross-cutting theme across all of the Research Teams supported through this funding opportunity. The cross-cutting themes of Knowledge Mobilization, Data and Biosamples, and Training and Capacity Development are required to be integrated within each funded Research Team. Additional considerations to be incorporated in Research Team activities, as appropriate, include Health Disparities and Equity, Diversity and Inclusion (EDI); Indigenous Peoples (First Nations, Inuit or Métis) Health and Wellness; Patient and Citizen Engagement; Lifecycle Approach; Leveraging Resources; and, Ethical, Legal and Social Considerations.
Each Research Team must designate an individual responsible for each cross-cutting theme (Theme Lead). Once funded, Research Teams will be expected to collaborate on cross-cutting theme activities. In addition to the Nominated Principal Applicant, Research Teams are to be interdisciplinary and include Early- and Mid- Career Investigator(s); trainee(s); a Knowledge User(s); Person(s) with Lived/Living Experience; and Indigenous communities (as appropriate).
In-Kind Partner Contributions
| Contact Information | Partner Role | Relevant Research Area(s) | Available in-kind resource(s) |
|---|---|---|---|
|
Paula Robeson, RN, BN, MScN |
Knowledge Mobilization | Children’s Health | Through the SPARK Impact consultation and support service for researchers, Children’s Healthcare Canada, is well-positioned to provide child health researchers applying for the team grant with a “free” initial consultation to inform the development of grant’s the Knowledge Mobilization plan and include the costs of additional supports from Children’s Healthcare Canada into their team grant applications. |
|
Paul Eckford, PhD |
Knowledge Mobilization Partner Patient Engagement |
Cystic Fibrosis | Cystic Fibrosis Canada operates Elevate, which is a CF community engagement tool to assist researchers reach CF community members for participation in surveys, engagement activities etc. CF Canada can also be engaged to assist with KM activities for relevant CF research, as the organization is highly engaged with the Canadian CF population and has direct relationships with the 40 CF clinics that service virtually all CF patients in Canada. CF Canada operates the CF patient data registry, which can be accessed (via an application process) for CF-relevant health data and trends. Finally, CF Canada operates CF CanACT, a network of sites across Canada for conducting CF-specific clinical trials. CF CanACT can be engaged for protocol reviews and site selection for relevant clinical trials arising from this initiative. CF Canada should be an important partner in any CF-relevant studies through this initiative. |
| Controlled Substances and Cannabis Branch Health Canada sp-licensing-cannabis-licences-sp@hc-sc.gc.ca |
Cannabis Regulator | Cannabis Legalization and Regulation | Amendments to the Cannabis Regulations were enacted in December 2022 to exempt non-therapeutic research on cannabis involving human participants from the clinical trial requirements under the Food and Drug Regulations, where that research is conducted under a cannabis research licence issued under the Cannabis Regulations. The amendments also include appropriate health and safety requirements to protect the participants. Health Canada serves to guide applicants seeking a cannabis research licence by sharing application requirements or answering questions about the Cannabis Act and its Regulations including the non-therapeutic research on cannabis framework. Health Canada also supports cannabis research licence holders with post licensing requests. |
| Scott Mitchell Manager, Knowledge Mobilization Canadian Thoracic Society 1000 Innovation Dr. Suite 500 Ottawa, ON, K2K 3E7 smitchell@cts-sct.ca |
Knowledge Mobilization Partner | Lung Health, Inflammation, Climate Change, Vaping |
CTS will support this initiative through knowledge dissemination and exchange within our membership and through our extensive knowledge mobilization avenues, networks, and partners in respiratory health. CTS is Canada’s national inter-professional specialty society and membership association for respirology, representing specialists, physicians, researchers, and health care professionals from a variety of disciplines working in respiratory health. We are recognized as an accredited provider of continuing professional development and a member-driven organization that develops and delivers education and knowledge translation activities that address gaps in knowledge and enhance competencies. CTS hosts the annual Canadian Respiratory Conference which brings together over 600 clinicians, scientists, researchers to share the latest updates in respiratory health care. |
Contact Information
To learn more about how your organization can partner on this initiative, please contact icrh-iscr@cihr-irsc.gc.ca.
- Date modified: